- New data show that the site management model reduces start-up times for a Phase II or Phase III clinical trial by up to nine months compared to UK public sites, representing billions in savings
- Panthera identifies likely patients seven months before public sector sites start planning their recruitment campaigns.
The loss of nine months patent on a blockbuster drug represents billions of dollars to pharmaceutical companies. Speeding up the initiation processes to start recruitment and enrol patients into a clinical trial is one of the easiest steps that a study sponsor can take.
Panthera Biopartners, a leading Site Management Organisation (SMO) in Western Europe, has analysed the various stages of start-up involved when initiating a clinical trial and compared the model to UK NHS public sites that still recruit most UK patients to commercial clinical trials.
Data from the National Institute of Health and Care Research (NIHR) show that the average time between public sector study site selection and the first patient recruited averages between 66 days to 93 days.[1] In addition to this, its latest figures, for Q1 2022, show that for 1,191 trials, only 51.4% of recruitment targets are being met.
The UK and the NHS has long been at the forefront of clinical research, and a valued partner for both pharmaceutical companies, academics, and patients alike. However, figures from the SMO model and directly from NHS show the differences in site selection and recruitment speed.
While the NHS do not break down figures for each phase of the study start up, Panthera is able to identify at which stage of the process the SMO model offers the most value versus the public sector.
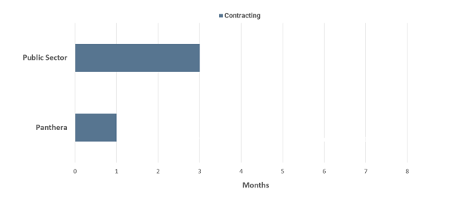
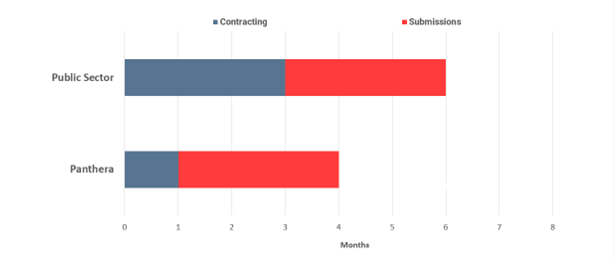
Contracting
- Public sector sites take on average three months to agree contract terms as lawyers and clinicians involved in the discussions have many other priorities.
- Panthera aims to agree contract terms in as short as one month as Panthera is entirely focused on ensuring that the contract terms are agreed as quickly as possible with no other distractions.
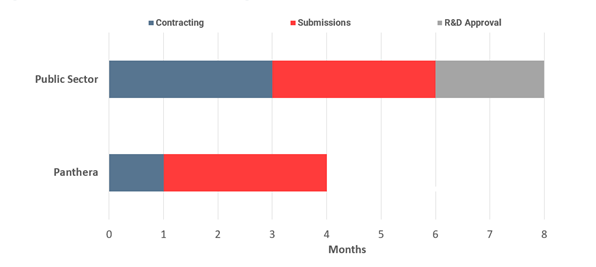
R&D approval
- All NHS public sector sites must submit a clinical trial protocol to their NHS trusts R&D committee for approval of the protocol. This takes on average two months.
- SMOs do not have to seek R&D approval.
Ethical approval
- Ethics approval in the UK takes between 30 and 35 days. There is no difference between the SMO model and public sector sites.
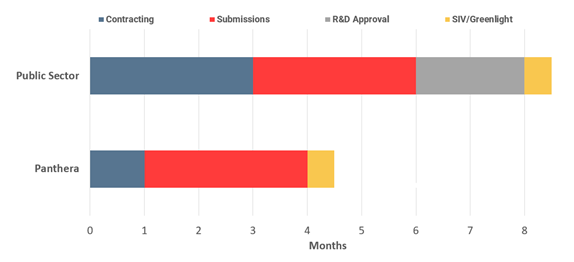
Site initiation visits
- Every clinical trial site must be inspected and validated by the CRO or pharma responsible for the clinical trial.
- Panthera prioritises the pharma or CRO and aims to have the initiation completed in two weeks
- Public sector sites may have many other calls on their time and availability and take on average four weeks for site initiation.
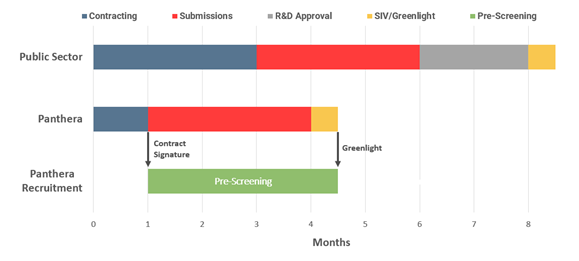
Green light to go forward
- Following all approvals and initiation visits, public sector sites start to recruit patients.
- After contract signature, Panthera starts pre-screening patients who may be applicable for the trial. The SMO model is already identifying likely patients > seven months before the public sector sites start planning their recruitment campaigns.
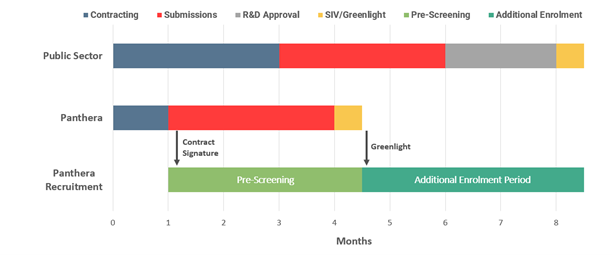
Additional recruitment
- In up to 75% of cases, clinical trials for Phase II and Phase III run late and erode the peak sales at the end of the patent life costing pharma billions of dollars.
- An SMO can boost recruitment numbers early to meet last patient-in deadlines
Panthera’s SMO model is applicable across general medicine as well as specialist areas such as oncology, NASH, neurology and vaccines.
